
They are so called because ions move toward the electrode of opposite charge. A cation is something that moves down ( Greek: κάτω pronounced kato, meaning "down") and an anion is something that moves up ( Greek: ano ἄνω, meaning "up"). The word ion was coined from Greek neuter present participle of ienai ( Greek: ἰέναι), meaning "to go". Ions are also created by chemical interactions, such as the dissolution of a salt in liquids, or by other means, such as passing a direct current through a conducting solution, dissolving an anode via ionization. 
In the case of physical ionization in a fluid (gas or liquid), "ion pairs" are created by spontaneous molecule collisions, where each generated pair consists of a free electron and a positive ion. Ions consisting of only a single atom are termed atomic or monatomic ions, while two or more atoms form molecular ions or polyatomic ions. Opposite electric charges are pulled towards one another by electrostatic force, so cations and anions attract each other and readily form ionic compounds. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons.Ī cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. ɒ n, - ən/) is an atom or molecule with a net electrical charge. Forming an ionic bond, Li and F become Li + and F − ions.Īn ion ( / ˈ aɪ. In this animated video tutorial, you will learn about ions and charges, how to assign charges to monoatomic and polyatomic ions, and the difference between ionic compounds and molecular compounds.For other uses, see Ion (disambiguation).Įlectron transfer between lithium (Li) and fluorine (F). Video tutorial – Ions, Polyatomic Ions, and Ionic Compounds You can read about naming ionic compounds, and naming covalent compounds. Learning how to properly name compounds like these is important. BeBr 2 – (berylliuim bromide) cation: Be 2+, anion: Br –.K 2S – (potassium sulfide) cation: K +, anion: S 2.Mg(OH) 2 – (magnesium hydroxide) cation: Mg 2+, anion: OH –.LiF – (lithium fluoride) cation: Li +, anion: F –.NaCl – (sodium chloride) cation: Na +, anion: Cl –.For example, in NaBr, sodium is the cation, while bromine is the anion. When writing the chemical formula of a compound, cation always comes before anion. Most metals (e.g., iron, lead, gold) form cations, whereas most nonmetals (e.g., oxygen, nitrogen, sulfur) form anions. In contrast, Group 17A, which consists of halogens, always forms anions. Group 1A and 2A of the periodic table, alkali metals and alkaline earth metals respectively, always form cations. Whether an atom forms a cation or an anion depends on its position on the periodic table. 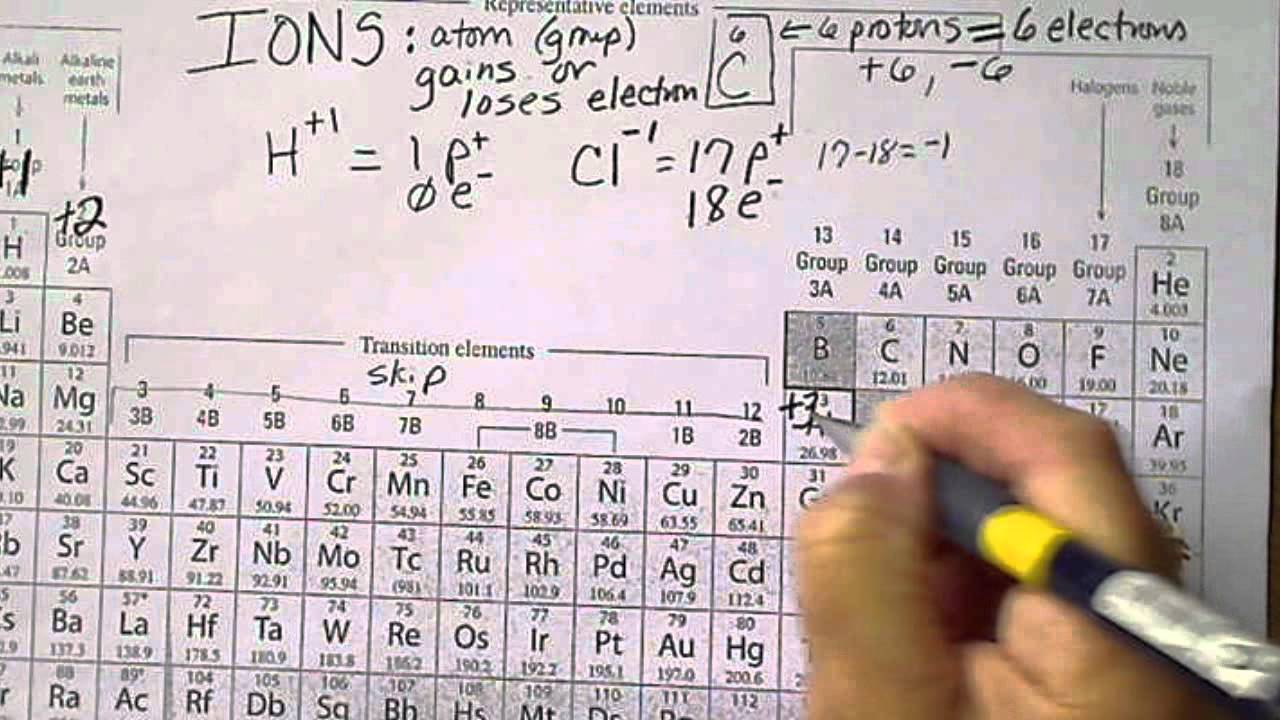
Predicting Cations and Anions based on the Periodic Table Ions like hydroxide, dichromate, sulfate, and ammonium that contain more than one type of element are called polyatomic ions and are discussed in more detail in another article. Because electrons are added to form an anion, the anion of an atom is bigger than the neutral atom.Įxamples of anions include the following: What is an anion? An anion is an ion that has gained one or more electrons, giving a net negative charge. Because one or more electrons are removed to form a cation, the cation of an atom is smaller than the neutral atom.Įxamples of cations include the following: What is a cation? A cation is an ion that has lost one or more electrons, giving a net positive charge. There are two types of ions: cations and anions.Ī cation has a net positive electrical charge, which means it has more protons than electrons.Īn anion has a net negative electrical charge, which means it has more electrons than protons. Ions are identified by a superscript that shows the sign and size of the electric charge – for example Ca +2. Ions form when atoms move into a more stable electron configuration. They contain different numbers of protons and electrons. The definition of an ion is a particle, atom, or molecule with an imbalance of electrical charge. Introduction to Ions, Cations and Anions What is an Ion? What are ions? In this tutorial, you will learn about the properties, differences, and examples of ions, cations and anions, as well as how to predict them based on their positions on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
